Understanding Medical Device Design
16/09/2021, hardwarebee
Medical electronics are a profitable market that is increasingly expanding, especially with the fast proliferation of embedded technologies and wearable devices. Medical design generally aims safety, effectiveness, automation and reliability, as possible errors may cause dangerous consequences to the user or patient. Although profitable, medical design is difficult, not only because the device must be as reliable as possible to guarantee user safety, but also due to the regulatory requirements that medical equipment must satisfy before market release. To avoid the frustration of having the project rejected after all the hard work, the designer must be aware of all the steps necessary to comply with the healthcare regulatory requirements, while keeping the device safe, effective and under the budget. This article provides an overview of some important parts of the medical design flow, focusing mostly on electronics for healthcare.
WHAT IS A MEDICAL DEVICE?
The requirements that a product must satisfy to be recognized as a medical device may vary from country to country. However, the FDA (Food and Drug Administration, the most important medical regulatory agency in the US) definition is the most applied around the globe. According to them, a medical device is defined as
- any equipment, instrument of apparatus that is intended to cure, mitigate, treat, diagnose or prevent a disease or condition, or
- any equipment, instrument of apparatus intended to modify, affect, or replace a function of the
- Either of the above must not achieve the intended function primarily through chemical or metabolic
Because this definition is broad, there are countless instruments in the market that can be classified as medical device: from bedpans and braces to pacemakers and electric wheelchairs. Some engineers may not even be aware that their product is a medical device before the market demands proper certifications. Therefore, it is extremely important to understand how medical regulation in the target country works before starting the design flow. Once it is clear that the product is a medical device, one should verify what class the device belongs to.

Figure 1 – Different Medical Devices
DEVICE CLASSIFICATION
The FDA defines 3 classes which, among other things, specify the type of premarketing submission/application that is required by the regulation. Medical device classes are defined according to the risk and nature of the product.
Class I
Class I devices are products that are not intended for use in life-support or sustaining applications, and do not present significant risk for the user. Therefore, the FDA certification process is much less restrict for this class, and most medical devices approved are Class I. Some examples are bandages, hospital beds and tongue depressors. Most Class I devices do not require FDA Pre-Market notification or approval before release, but should still comply with the FDA General Controls during the design and manufacturing.
Class II
Class II devices are more risky than the Class I. Typically, they require prolonged contact with the patient body, and can be invasive. Therefore, there is substantial risky to the user if misused or during malfunctioning. In these cases, the FDA General Controls are not enough to ensure safety and effectiveness. Some classic examples are contact lenses, catheters and syringes. Besides special controls, which are different for each device, most Class II devices must be approved through Pre-Market Notification process, also known as 510(k).
Class III
Any device that satisfies one of the following is considered a Class III device:
- It operates a life-support or life-sustain function;
- It is an implant;
- Presents a significant risk of injury or illness to the
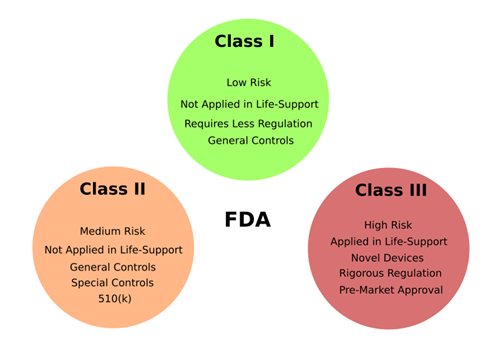
Figure 2 – Risk Classes
REGULATION
After verifying the class of the medical device, a careful designer should check the regulation regarding that class. As explaining previously, different classes require different types of approval before market release. Also, the FDA defines a list of classification panels, grouping devices into several categories according to the function it performs.
Classification Panels
The FDA divided over 1700 types of devices into 16 categories, called classification panels, such as ”cardiovascular”, ”neurology” and ”physical medicine”. Each classification panel has its general description, the intended use of the device, the class (I, II or III) and the regulation (controls and marketing approval requirements). Therefore, finding out which panel the device belongs to is a crucial part of the design flow.
General and Special Controls
General controls are the basic set of rules that is common to all medical devices. It includes, among others, provisions concerning adulteration, misbranding, notifications and good practices. It is the only requirement for most of Class I devices. Special controls, on the other hand, are device specific provisions that are added to the general controls, when the latter are not sufficient (Class II devices). Specific controls typically include post-market surveillance, pre-market data requirements and additional guidelines.
Pre-market Notification 510(k)
Pre-market notification, or ”510(k)”, is an additional requirement for most Class II devices, although the FDA can demand it for some Class I and Class III products. The 510(k) is a technical description of the device that includes detailed technical and functional information, especially concerning safety and effectiveness. The FDA uses the 510(k) document to check if the device is substantially equivalent to another medical device already approved in the market. Therefore, in order to obtain a 510(k) clearance, the device should be similar to an already existing device.
Pre-market Approval (PMA)
Before marketing release, most Class III devices must obtain a PMA certification. PMA is regulated under the Title 21 Code of Federal Regulations (CFR), and any device that fails it cannot be marketed legally. Different from the 510(K), the PMA application is a scientific study of the device, which essentially proves the safety and effectiveness of the product. Therefore, the scientific method should be rigorously applied. Clinical trials are typically required, which makes the process very time and resource consuming. Also, every laboratory test, such as in vitro analysis, animal models and biocompatibility trials, should be reported. The FDA provides several technical guidelines and standards to orient designers during non-clinical and clinical tests, according to the device type. Clinical trials must, of course, comply with ethical and scientific regulations.
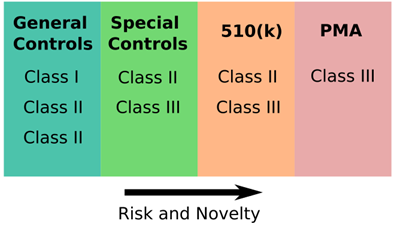
Figure 3 – Pre-Market Requirements for Device Class
ELECTRONICS APPLICATIONS AND REQUIREMENTS
Medical devices are a broad market, which includes both mechanical and electrical instruments and machinery. Here, we focus on medical applications involving primarily electronics, especially the ones applying embedded systems, to emphasize how the medical market is growing towards automated, small and integrated systems, moving closer and closer to the internet of things (IoT).
Diagnostic Equipment
This is the most recognizable type of device and usually the one that comes to mind when talking about medical equipment. Diagnosis devices include the classic X-ray machine, the ultrasound scan, and the magnetic resonance imaging (MRI) system, among others. Any device that measures a body quantity, which can be used to diagnose a disease or condition, falls into this category. This type of equipment must be very precise, reliable and robust, in order to give the correct answer independent on the environmental conditions. However, they are not limited to large and bulky devices. In fact, small and portable diagnostic tools are becoming more desirable, especially in remote areas with low resources. One example is using bioimpedance sensors for skin cancer diagnostics, which uses the difference between the electric impedance of healthy and tumorous cells to identify cancer.
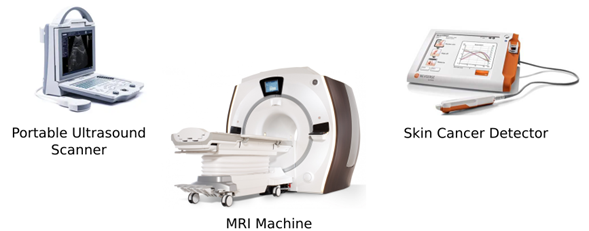
Figure 4 – Diagnostic Equipment
Treatment Machinery
Similar to diagnostic devices, this category typically consists of heavy and bulky equipment, often made of many moving parts working in a controlled environment. However, these machines are intended to treat or mitigate a disease or condition. Some examples are: hemodialysis machines, respirators, Extracorporeal Membrane Oxygenation (ECMO) systems, etc. They are typically applied in intensive care units (ICU), and should be extremely reliable and robust, because any error can result in patient harm or even death. Treatment devices also require several redundancy and fail-safe systems, which further increases its volume. Because of its invasive nature, it is difficult to develop small and portable treatment devices, but not impossible: for instance, automated insulin delivery systems for diabetics are a “hot trend” in the medical research community.

Figure 5 – Treatment Machinery
Assistive Technology
Any equipment or instrument implemented to increase, maintain or improve any functional capabilities of people with disabilities is an assistive technology (AT) device. Therefore, this is a broad field, that includes both physical, mental, temporary and permanent disabilities. One famous example is the electrical wheelchair, but AT includes hearing aid devices, sonar sensors for the blind, fall prevention systems and electronic prostheses. Different from the previous categories, AT typically does not require bulky systems. Instead, both functional, practical and aesthetic sides of the device should be carefully designed. This is because AT devices are typically used in the daily basis, so it should interfere as little as possible with the life style of the user, otherwise it will probably be rejected by the market. Embedded systems, especially using portable and wearable electronics, are broadly applied in AT.

Figure 6 – Assistive Technology
Implants
Implants are devices that are installed inside the user body, operating from within. The implant can be used to perform a function, such as a pacemaker, or to measure some quantity inside the body. There are three major problems with implants: the first is the installation process involves a surgery, which is almost always problematic to the patient. The second one is that only highly biocompatible materials can be in direct contact with the living tissue, which limits the manufacturing process significantly. Finally, there is the energetic problem: how to charge an implant without needing to perform a surgery? Concerning the last limitation, energy harvesting and wireless power transmission are exciting fields currently being investigated by the medical community. If technology can surpass these three bottlenecks, the medical implants market can grow exponentially, as these devices are able to operate directly attached to the tissue of interested, which yields an effective and ”invisible” performance.
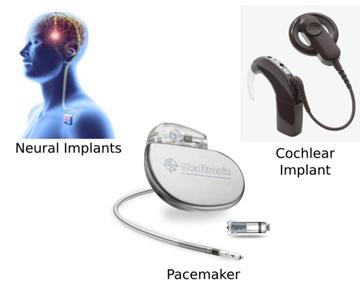
Figure 7 – Electronic Implants






